|
12/5/2023 0 Comments Dead space in lung 
Increased ventilation will increase tidal volume and thus increase your minute ventilation.Hint: No, residual volume and dead space volume are not synonymous. Lung units with high V/Q quotients, have high ventilation (decreasing pCO2 and increasing O2). Abnormal breathing PV = 150 x 40 = 6000mL/min. PV = TV x RR and AV = (TV - Dead space) x RR Normal: PV = 500 x 12= 6000mL/min. Dead space also depends on the size and posture of the patient. Therefore your dead space increases dramatically. Your pulmonary ventilation (total exchange of air between the lungs and ambient air-minute ventilation) will be 6000mL/min but your alveolar ventilation (total exchange of air that reaches the alveoli and is available for gaseous exchange with the blood per unit time) will be 0. If you breathe rapidly and shallow, lets say a TV of 150mL and a respiratory rate of 40/min. An increased respiratory rate will increase the volume of the dead space. Physiological dead space is the volume of gas that does not eliminate CO2. At the end of inspiration, the chest wall recoils and pulls lungs back to original position Anatomical dead space is 2mL/kg (150mL). due to thickening of the blood-gas barrier).Ĥ. 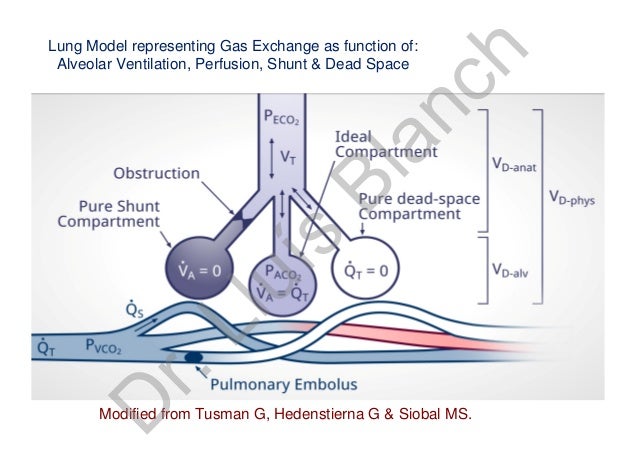
Depending on certain circumstance, O2 can be perfusion limited (normal) or diffusion limited (abnormal), e.g. It is therefore perfusion limited as the amount taken up by the blood depends entirely on the amount of available blood flow and not at all on the diffusion properties of the blood-gas barrier. 
NO is very insoluble in blood and quickly forms a partial pressure (back pressure). It does not form a partial pressure in the blood due to its extremely high solubility in blood (no appreciable back pressure develops). it is not dependent on the amount of blood available but rather only on the diffusion properties of the blood-gas barrier). CO is used for measurements because its uptake is diffusion limited (i.e. This states that the rate of transfer of a gas through a sheet of tissues is proportional to the tissue area and the difference in gas partial pressure between the two sides, and inversely proportional to the tissue thickness. Carbon monoxide is diffusion limited Diffusion through tissues is described by Fick's law (there is no Fowler's law). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
